|
|
|
There is no dye in the colors brought out in titanium, niobium, or tantalum.
|
Okay, so we know that a clear layer on metal is like a mirror (or vice-versa).
There are really 2 reflections.
So, if a light wave of a single frequency hits the mirror, it reflects once from the clear part,
and once from the metal. When they come out, they run into each other. What happens?
Right. They interfere with each other. If the dielectric is thick enough, like several wavelengths of light or more,
nothing noticeable happens. All the interesting effects are statistically cancelled out.
However, if the distance the light has to travel through the dielectric to bounce off of the metal
and back up to where it hits the first reflection is between ½ and 2 or so times the wavelength, then
the interference gets interesting. If, for example, the total length (twice the thickness) is about half the length of the
wavelength of red light, then it is also about the full wavelength of blue light. So, blue light will be reinforced, and red will be cancelled!
Since white is made up of all colors, the blue end of the rainbow comes back out, and the red gets lost.
Voila! Color. Maybe this illustration will help:
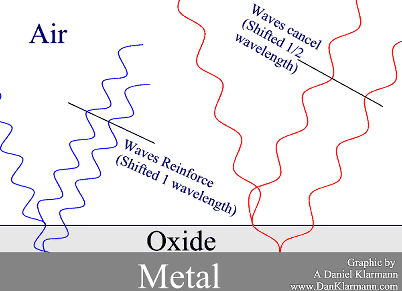
The shorter blue wavelength reflects of the surface, and off the metal.
The interior wave travels a full wavelength farther than the
first reflection, so they reinforce.
The longer red wave travels the same distance, but that's only ½ of its wavelength, so it cancels out.
When all the colors which are in white bounce off this double layer, what you see is blue.
bronze (the blue half of the spectrum is cancelled, and nothing else effected)
blues (red-green is cancelled)
blue-white (almost balanced, but still missing some red)
yellow (cancelling blue, the second time around. This is the practical high-end for heat coloring)
magenta (also called purple, cancelling green, leaving blue and red)
cyan (cancelling red more narrowly, leaving bue and green
green (cancelling both ends, keeping the narrow green band. Hard to get in Titanium, easier with Niobium.)
| Please use this forum post to ask me a question. |
Back to my Unique titanium jewelry